Suzuki reaction Monitoring using Compact Mass Spectrometry with the Plate Express TLC Plate Reader
Author: Advion, Inc.
Introduction
Thin layer chromatography (TLC) is used in many organic synthetic and medical laboratories because it is a simple, cost-effective technique that provides chemists with critical information about their synthetic reactions. Structural characterization of the analytes by TLC is not possible by optical  methods such as UV or ELSD. Typically, characterization is performed by GC/MS or LC/MS using sample preparation techniques which involves scraping the TLC spot of interest, extraction using suitable solvents, concentration, and then reconstitution in MS appropriate solvents.
methods such as UV or ELSD. Typically, characterization is performed by GC/MS or LC/MS using sample preparation techniques which involves scraping the TLC spot of interest, extraction using suitable solvents, concentration, and then reconstitution in MS appropriate solvents.
This application note presents an on-line TLC/CMS technique using the Advion expression® CMS and Advion Plate Express® to provide compound structural information without sample preparation after TLC separation. A Suzuki reaction for the synthesis of 4-aminobiphenyl will be demonstrated. The online TLC/CMS technique provides rapid and accurate determination of reaction mixture components without the need for off-line TLC sample preparation procedures.
Method
Reactants A and B were mixed at equimolar amounts in a round-bottom reaction flask and stirred at room temperature. 2 mL aliquots were transferred from the flask and spotted onto a Merck TLC Silica gel 60 F254 plate (10 x 10cm). Chemicals were purchased from Sigma-Aldrich with a purity greater than 99%. MS solvent was LC/MS grade.
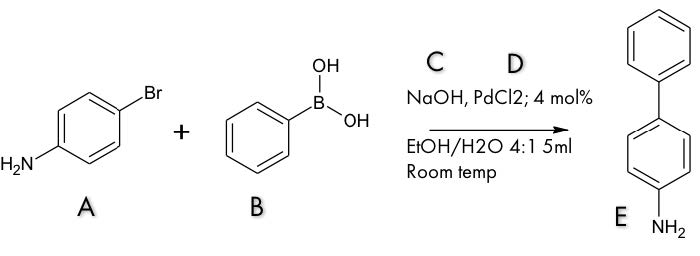
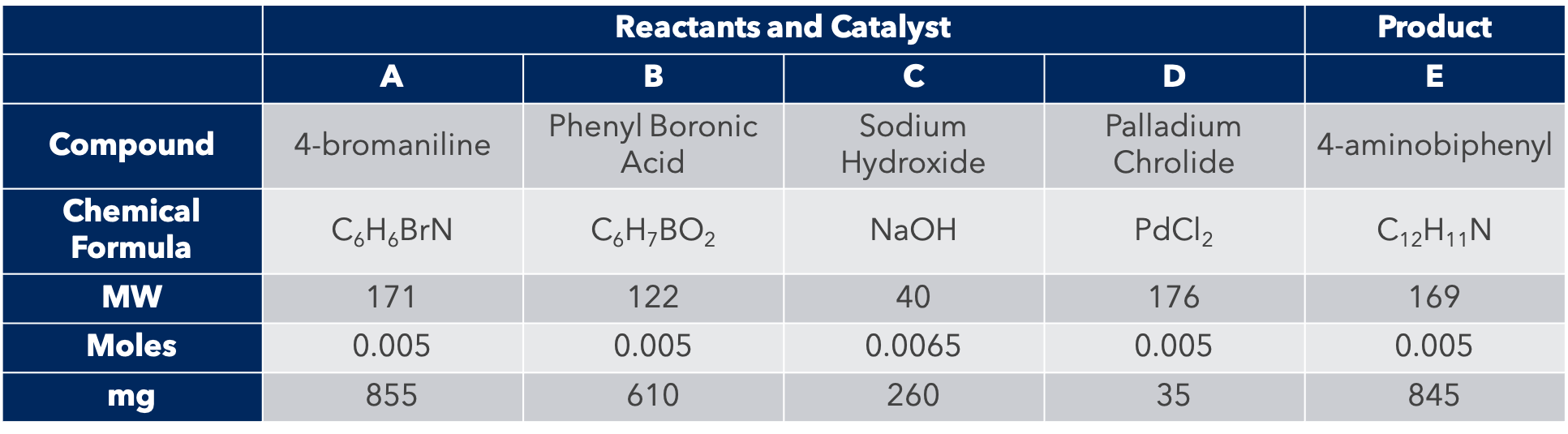
Figure 2. Chemical structure and information of reactants and catalysts for the Suzuki reaction synthesis of 4-aminobiphenyl.
TLC Plate Preparation
Merck TLC Silica gel 60 F254 on Aluminum, 20 x 20 cm – cut to10 x 10 cm. TLC plates were baked at 100°C for 10 minutes to displace moisture. A 2 uL aliquot of the reaction mixture was spotted onto the TLC plate at the position of 1 cm away from the bottom. The TLC plate was then baked at 80°C for 5 min. 30 mL Benzene was added to a TLC plate developing chamber at the level of 0.5 cm. The developing chamber was sealed with a glass lid for 30 min. The separation was stopped when the development solvent front reached a position, which was 1 cm away from the top of the TLC plate.
The analytes (reactant and product) on the developed plate were observed under UV at 254 nm (Figure 3). The Rf value of the product (4-aminobiphenyl) was 0.1; the Rf of the reactant (4-bromaniline) was 0.15.

Figure 3. The developed TLC plate with Suzuki reaction mixture.
TLC/CMS Method
The TLC/CMS analysis of the Suzuki reaction mixture at different reaction times was performed on the Advion expression CMS and Plate Express. A solvent composed of 0.1% formic in methanol was used for the elution of the analytes from the TLC plate. The eluted analytes were directed to the CMS for aquisition of the corresponding mass spectra for the reactants and products.
Results
The mass spectra of the reactant and product are shown in Figure 4. The [M+H]+ for 4-bromoaniline was observed at m/z 171.9 (79Br) and 173.9 (81Br). Methanol adducts were also detected at m/z 204.0 and 206.0 (Figure 4A).
The [M+H]+ for 4-aminobiphenyl was observed at m/z 170.1 with a methanol adduct at m/z 202.1 (Notice the absence of the bromine doublet peaks in the non-brominated product of p-aminobiphenyl, Figure 4B).
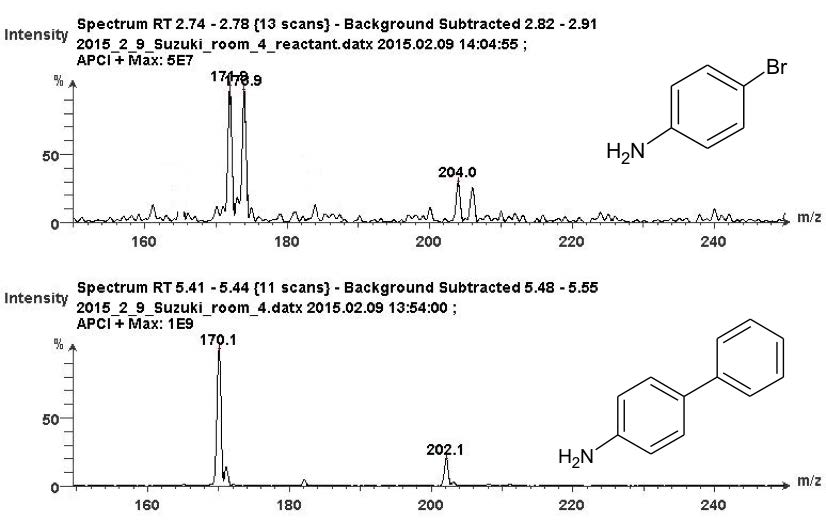
Figure 4. TLC/CMS analysis of reactant and product. (Top) The mass spectrum of protonated 4-bromoanaline (reactant) at the m/z 171.9 and 173.9. (Bottom) The mass spectrum of portonated 4-aminobiphenyl (product) at m/z 170.1.
The extracted ion current of the product ion at m/z 170.1 (protonated 4-amionbiphenyl) was monitored over different reaction times from 0 to 180 minutes (Figure 5A). The mass spectrum of the product ion at 120 min is shown in Figure 5B. The extracted ion current (XIC) of the reactant ion at the m/z 171.9 (protonated 4-bromaniline(79Br)) is shown in Figure 5C. The intensity of the m/z 171.9 decreased over the course of the reaction with respect to the product ion at the m/z 170.1 The reactant ion at the m/z 171.9 was no longer detected at 180 minutes, indicating the reaction was complete.

Figure 5. (A) XIC of product ion (protonated 4-aminobiphenyl). (B) Mass spectrum of the product ion at 120 min. (C) XIC of the reactant ion (protonated 4-bromonailine). (D) Mass spectrum of the reactant ions at 15 minutes.
Summary
- The Advion expression CMS coupled with the Advion Plate Express offers a simple and fast technique to monitor a Suzuki reaction for the synthesis of 4-aminobiphenyl.
- The Advion TLC/CMS system allows the synthetic chemist to monitor the reaction in real-time by evaluating the mass spectra for structural information (i.e., relative intensity of reactants vs. product) directly from the TLC plate.
- The compact size allows it to fit into space-limited labs for direct access and immediate results for chemists requiring mass confirmation, reaction monitoring, quality control, and purity analysis.
