Introduction
Environmental contaminants generated by human or industrial activities often find their way to the soil via runoff waters or deposition from the air. These contaminants can be taken up by plants and move up the food chain leading to potentially significant impacts on human and animal health. Therefore, it is not only important to monitor the levels of essential nutrients in the soil that are key for healthy plant growth, but it is also imperative that the levels of contaminants are monitored.
In this application note we present a method for routine analysis of 21 elements using the SOLATION® Inductively Coupled Plasma Mass Spectrometer (ICP-MS). A group of unknown soil samples and a CRM were digested using EPA 3051a and analyzed according to method 6020a requirements.
Experiment
Reagents and Materials
• Nitric acid (Aristar Plus, trace metal grade)
• Hydrochloric acid (Aristar Plus, trace metal grade)
• Water, type 1 (18.2 MΩ, Elga point of use system or equivalent)
• NIST CRM2706 “New Jersey Soil, Organics and Trace Elements”
• Spex ‘CL-ICV-1’ multi-element solution
• Aluminum standard solution (1000 μg/ml, Claritas ppt grade)
Instrumentation
1. Anton Paar Multiwave 5000 with the 20SVT50 rotor (20 position, 50mL vessels that vent at 40 bar (580 psi)
2. OKF high speed multi-function grinder
3. Advion Interchim Scientific SOLATION® ICP-MS
Standards
Calibration standards were prepared in the same acid proportions as the digested samples (9mL HNO3+ 3mL HCl, or 3:1). One liter of 3% HNO3+ 1% HCl was made as the diluent for standards, for the final dilution of samples, and to use as a calibration blank.
Standards were made using the Spex multi-element solution ‘CL-ICV-1’ and the single element Aluminum standard. Aluminum was added separately to the mix to account for the high levels of this element in soil.
Samples and Preparation
Four soil samples were dried at 60°C overnight, then finely ground using an OKF high speed multi-function grinder to make a homogeneous mixture. As per EPA method 3051a “Microwave assisted acid digestion of sediments, sludges, and soils”, 0.5 g of each sample were transferred to microwave vessels and mixed with 9mL nitric and 3mL hydrochloric acids. The vessels were then capped and run using the method outlined in Table 1. After digestion the samples were filtered, brought to volume with deionized water in a 50mL volumetric. A 1.0mL aliquot was then diluted to a final volume of 50mL with the prepared diluent for a nominal final dilution of 5,000x depending on the initial sample weight.
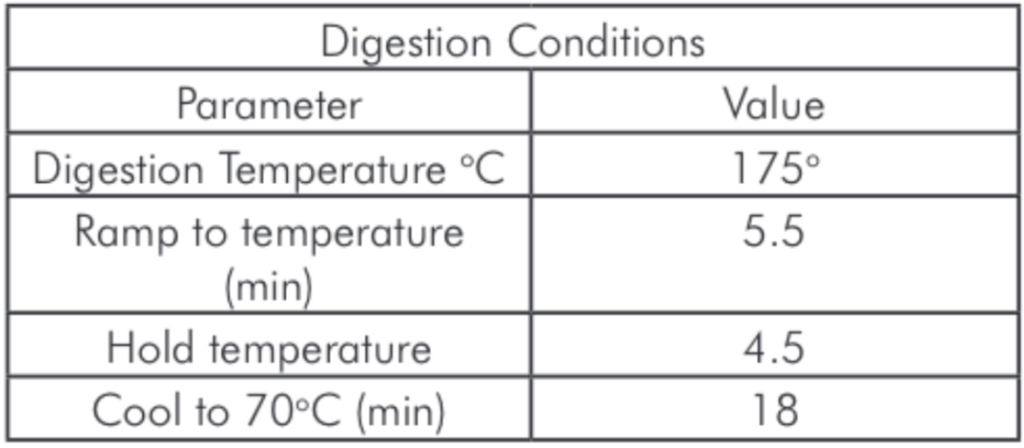 Table 1: Microwave Digestion Program.
Table 1: Microwave Digestion Program.
For QC purposes the four unknown soil samples were prepared as a sample, duplicate, and spike. They were independently digested where the first two were used to compare the repeatability of the sample preparation, while the third one was spiked prior to the digestion to establish analyte recovery of the digestion procedure.
To verify the accuracy of the results, we included the standard reference material, NIST 2706 “New Jersey soil, organics and trace elements”, which includes certified values for all analytes reported in this study.
The samples were analyzed using a SOLATION® ICP-MS. The SOLATION® instrument configuration for this analysis was a cyclonic spray chamber with a Micromist® concentric nebulizer and a one-piece torch. Ni sampler and skimmer cones were used throughout the study. The plasma operating parameters were:
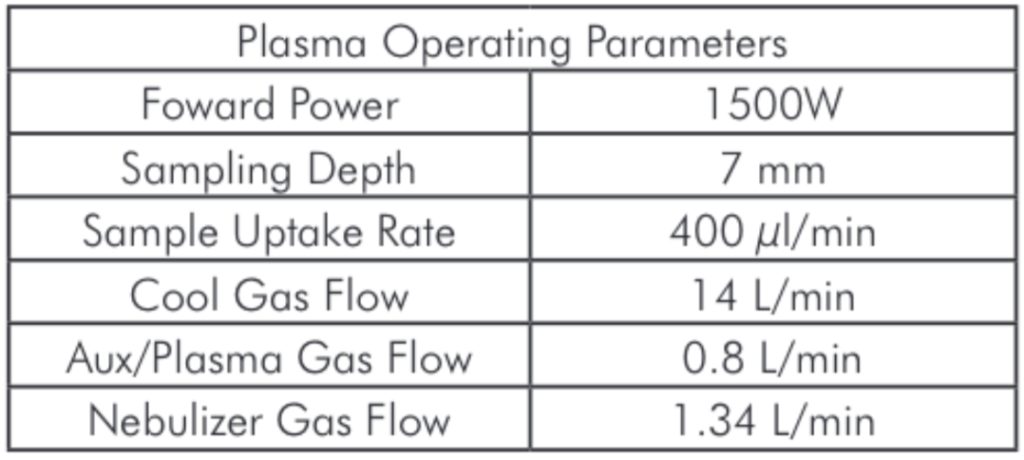 Table 2: Plasma Operating Parameters.
Table 2: Plasma Operating Parameters.
ICP-MS Method
Integral to the SOLATION® ICP-MS is an octupole collision cell that is used for addressing interferences from polyatomic ions, especially for the transition metal elements. It is critical for robust and routine trace element analysis that the octupole cell does not become contaminated which could cause drift and unnecessary downtime. Therefore, the ion path of the SOLATION® ICP-MS was designed to have the collision cell out of the direct line of the plasma. Ions passing through the interface are directed through a 90 ̊ turn and focused onto the entrance of the octupole using a quadrupole deflector (QD). Light and neutral particles continue through the QD and away from the cell.
The collision cell in the SOLATION® ICP-MS can be operated in “He Gas” mode in which the cell is filled with He to act as a collision gas, or in “No Gas” mode in which the cell is empty. The “He Gas” mode is used for isotopes subject to polyatomic interferences while the “No Gas” mode is used for the rest of the isotopes. The rapid switching between “He Gas” and “No Gas” modes on the SOLATION® (< 5 sec) ensures that analytical runs can be kept short, thereby improving productivity.
The helium flow used for “He Gas” mode in this application was 6 ml/min. Table 3 lists the elements used for this analysis and their isotopes, and the mode used for each.
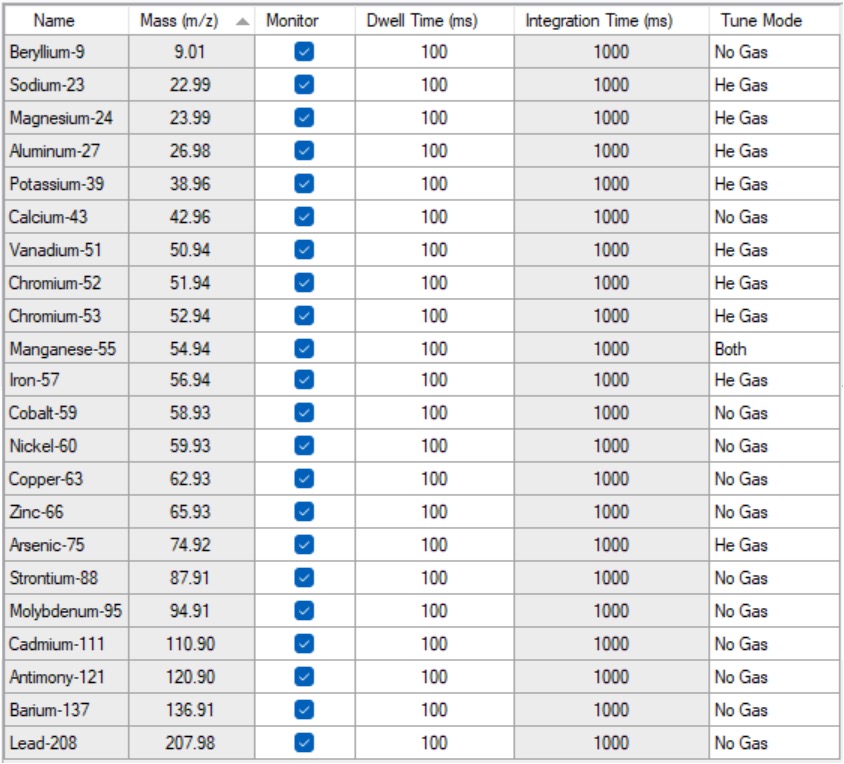 Table 3: A list of the elements included in this study together with their isotopes and the gas mode used for the analysis.
Table 3: A list of the elements included in this study together with their isotopes and the gas mode used for the analysis.
Results and Discussion
The results summarized in Figure 1 show excellent agreement between the measured data for CRM2706 and the reported extracted levels for these elements. A slightly higher recovery was observed for K and Al, possibly due to variability in the extraction efficiency of this digestion method.
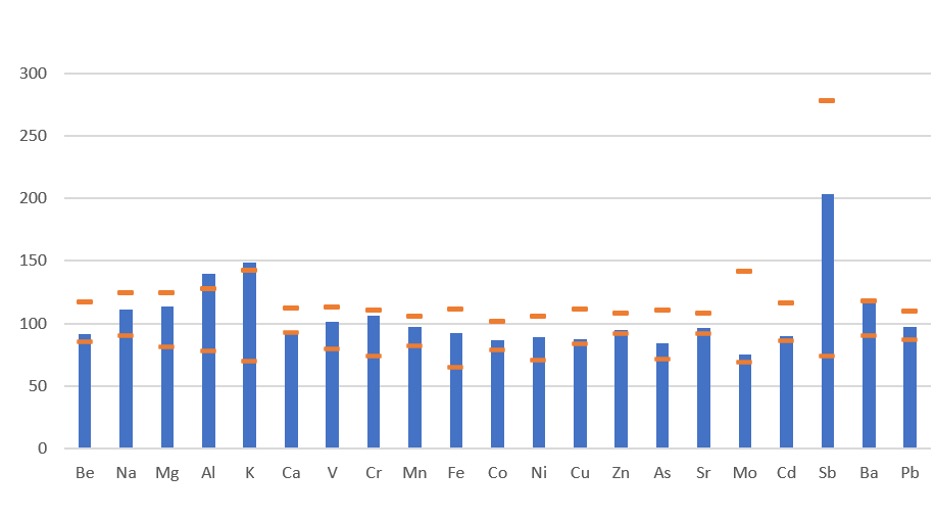 Figure 1: Certified reference material recovery data.
Figure 1: Certified reference material recovery data.

As shown in Table 4 spike recoveries averaged between 75% and 125% for all elements, with the exception of Al; This was likely due to the small size of the spike compared to levels of Al in the samples. Included in the same table are the results from the duplicate digestions/analyses for these elements. On average, the duplicates were less than 20% apart with most elements showing excellent repeatability of <5%.
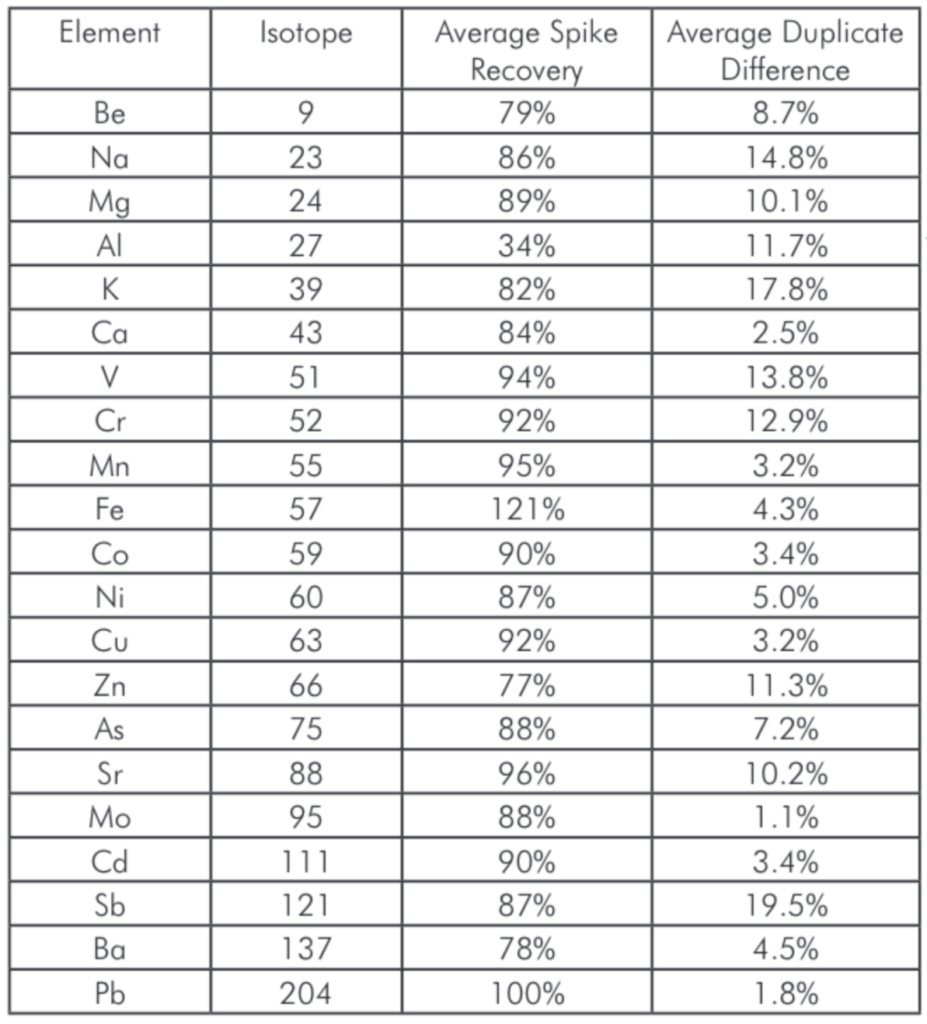 Table 4: Average spike recoveries and duplicate repeatability for the various samples.
Table 4: Average spike recoveries and duplicate repeatability for the various samples.
Summary
In this application brief we report on the analysis of trace elements in soil using the Advion Interchim Scientific SOLATION® ICP-MS. Excellent recoveries were observed for both spiked samples and CRMs. The combination of the quadrupole deflector and the collision cell minimizes drift and ensures accuracy and precision over time. The reported method benefits from the fast collision cell gas switching capabilities of the SOLATION® to analyze a wide range of elements in soil for rapid, accurate and reproducible results.