University of Cambridge
Abstract
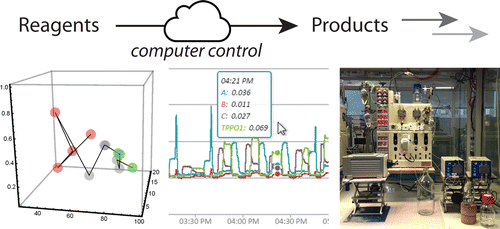
We have developed a modular software system that enables researchers to monitor and control chemical reactions via the Internet, using any device from any location in the world. It facilitates the automation of synthetic procedures and is able to autonomously self-optimize reaction parameters to find the best conditions meeting customizable, multicomponent optimization functions. In this report, we demonstrate its utility as applied to reaction automation to maximize the output from a fixed volume of catalyst. We also showcase its ability to optimize a three-dimensional heterogeneous catalytic reaction and a five-dimensional Appel reaction against various target functions.
In this publication, the Advion expression Compact Mass Spectrometer (CMS) was paired with a flow reaction system for simple reaction monitoring.
This note covers work carried out at Leeds University with co-workers from Durham University in the area of flow chemical synthesis using the expression CMS (Advion, Ithaca, NY). Two different reactions are described.

Q: WHAT IS THE FOCUS OF YOUR LAB’S RESEARCH?
A: The Liverpool School of Tropical Medicine (LSTM) is dedicated to research and teaching on tropical diseases. We are helping improve the health of some of the poorest people in the world by carrying out scientific research and developing treatments that will directly benefit them.
Specifically, I work in the department of parasitology, where our focus is on the key tropical parasites such as malaria, TB and filarial worms.
Q: WHY DID YOU INCORPORATE THE EXPRESSION CMS INTO YOUR LABORATORY?
A: The expression was originally purchased to save us from a 15-minute walk to get simple MS data at the University’s main analytical service. Also, it is compact and very easy to use, you do not need to be an expert in MS to generate very useful and meaningful data.
Q: HOW DO YOU USE THE EXPRESSION CMS?
A: We use the expression in the direct injection configuration for checking whether reactions have worked and whether the next purification and/or reaction steps are worth proceeding.
Before we got the expression, we either had to walk across campus to use the central service or continue without any knowledge as to whether our reactions had worked. The expression saves us time and boosts our overall research performance.
I am also planning to use it for some simple metabolism studies in the near future.
The analytical verification of compounds in organic syntheses is a challenging task. The sample is often directly taken out of the reaction mixture and therefore contains many impurities and matrix components. As a result, low-level sample preparation combined with a direct analysis method is highly beneficial. TLC/MS (thin layer chromatography – mass spectrometry) is such an analytical technique.
Co-authored with Merck Millipore in Darmstadt, Germany, this application note demonstrates a normal phase TLC/CMS technique for the rapid determination of 4-aminobiphenyl in its reaction mixture.
For the application note on the TLC/CMS determination of 4-aminobiphenyl in reverse phase, click here.
The analytical verification of compounds in organic syntheses is a challenging task. The sample is often directly taken out of the reaction mixture and therefore contains many impurities and matrix components. As a result, low-level sample preparation combined with a direct analysis method is highly beneficial. TLC/MS (thin layer chromatography – mass spectrometry) is such an analytical technique.
Co-authored with Merck Millipore in Darmstadt, Germany, this application note demonstrates a reverse phase TLC/CMS technique for the rapid determination of 4-aminobiphenyl in its reaction mixture.
For the application note on the TLC/CMS determination of 4-aminobiphenyl in normal phase, click here.

Q: WHAT IS THE FOCUS OF YOUR LAB’S RESEARCH?
A: As part of the Archibald research group our main research area is developing radiopharmaceutical compounds for medical imaging and targeted protein binding molecules for therapeutic applications.
Specifically, I am working on synthesising radiotracers to target the CXCR4 chemokine receptor which has been shown to be overexpressed in 23 different types of cancer.
Q: WHY DID YOU INCORPORATE THE EXPRESSION CMS INTO YOUR LABORATORY?
A: The expression CMS was purchased as we are a large research group and it is more cost and time effective to be able to carry out rapid MS analysis in situ for some sample and to use the University’s analytical service only for more complex analyses that are less time sensitive. The expression CMS fits nicely onto the bench top, performs all the functions needed for general day to day use, and is easy to use and maintain.
Q: HOW DO YOU USE THE EXPRESSION CMS?
A: We use the expression CMS to monitor chemical reactions, check for by-products and gauge the purity of our products. We have the TLC-MS interface which is extremely useful in identifying compounds straight from the TLC plate. Personally, I have used the expression CMS to examine fractions from a column to identify exactly when my product was being eluted, which would not have been possible previously. Purchasing the expression CMS has undoubtedly saved our group a great deal of time and has helped advance our research.

Q: WHAT IS THE FOCUS OF YOUR LAB’S RESEARCH?
A: The Drug Discovery Unit (DDU), based at the University of Dundee, is an independent, fully integrated Drug Discovery Group that works across multiple disease areas. We are active in areas of unmet medical need and bridge the gap between academic research and industrial drug discovery. Our staff are multi-disciplinary and come from both Academic and Industrial backgrounds.
Q: WHY DID YOU INCORPORATE THE EXPRESSION CMS INTO YOUR LABORATORY?
A: We needed a mass spectrometer that was easy to install, operate and move as well as available at a reasonable price. We have had the instrument for over a year now and are very happy with it.
Q: HOW DO YOU USE THE EXPRESSION CMS?
A: We have the expression linked to an HPLC instrument and it is used as an open access system. The combination of UV purity data along with MS confirmation is incredibly powerful for our chemists. The instrument is networked so our chemists can then process and review data from their desks.
Q: WHAT IS THE FOCUS OF YOUR LAB’S RESEARCH?
A: At SynZeal Research Pvt Ltd, with state of the art synthesis laboratory, we are focused on custom synthesis of high purity secondary reference standards of API Impurities and metabolites. SynZeal also provides chemistry research services and products to synergize the drug discovery and development.
Q: WHAT PREVIOUS WORKFLOW CHALLENGES DID YOU EXPERIENCE?
A: The major challenge to our workflow is to have fast decision on success or failure of targeted chemical transformation to decide further course of action and to complete the projects on time.
Q: WHY DID YOU INCORPORATE THE EXPRESSION CMS INTO YOUR LABORATORY?
A: We selected expression CMS as one of the affordable alternative for Mass Spectroscopy which can be operated even by an inexperienced person in mass spectrometry. Our productivity was increased many fold after installation of this system.
There have been a few reports on thin layer chromatography/mass spectrometry (TLC/MS) and thin layer chromatography/tandem mass spectrometry (TLC/MS/MS). The merits of TLC/MS (i.e., without MS/MS or high-resolution mass measurement) include lower-cost mass spectrometers, ease of operation and molecular weight (MW) information when applicable. TLC/MS/MS offers additional benefits, but requires more expensive instrumentation and more challenging operational requirements.
This article describes the direct coupling of TLC plates or other planar surfaces to a compact, single-quadrupole mass spectrometer that can provide routine electrospray ionization (ESI) or atmospheric pressure chemical ionization (APCI) in both the positive and negative ionization modes.
This application note presents an online thin-layer chromatography (TLC)/CMS technique using the Advion Interchim Scientific expression® compact mass spectrometer (CMS) and Plate Express™ interface to provide compound structural information without sample preparation after TLC separation. A Suzuki reaction for the synthesis of 4-aminobiphenyl will be demonstrated. The online TLC/CMS technique provides rapid and accurate determination of reaction mixture components without the need for off-line TLC sample preparation procedures.





